
The color code used is:

| Principle Quantum Number | Orbital | Orientation | ||||||
| 1 | s | |||||||
| 2 | s | |||||||
| p | 2px | 2py | 2pz | |||||
| 3 | s | |||||||
| p | 3px | 3py | 3pz | |||||
| d | 3dxy | 3dxz | 3dyz | 3dz2 | 3dx2-y2 | |||
| 4 | s | |||||||
| p | 4px | 4py | 4pz | |||||
| d | 3dxy | 3dxz | 3dyz | 3dz2 | 3dx2-y2 | |||
| f | 4fx (x2-3y3) | 4fxyz | 4fxz2 | 4fy (3x2-y2) | 4fyz2 | 4fz3 | 4fz (x2-y2) | |
The color code for the probability is: 
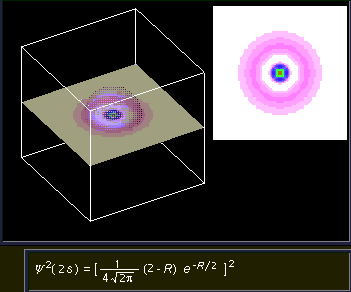
2s orbital. This image shows a slice of the 2s orbital that includes the spherical node (represented by the white circle of zero probability) in the 2s orbital.
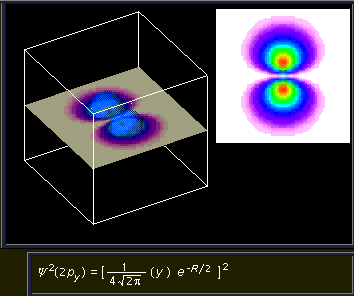
2py orbital. This image shows the intensity of the two nodes of a 2p orbital. Notice the planar node cutting through the center.
| Isosurface | (Local) |
| Slicing | (Local) |
| Volume Rendering | (Local) |
| 2s | (Local) |
| 2px | (Local) |
| 3px | (Local) |
| 3dxz | (Local) |
Please send any comments, corrections, or suggestions to svanbram@science.widener.edu.
This page has been accessed
times since 1/5 /96 .
Last Updated Sunday, November 03, 1996 11:19:05