Atoms & Elements Problem Set
This problem set was developed by S.E. Van Bramer for Chemistry 145 at Widener University.
- How many protons, neutrons and electrons are present in one atom of the following:
- 1H
- 2H
- 3H
- 13N
- 14N
- 15N
- 79Br
- 81Br1-
- 262Bh
- What is the Atomic Weight of chlorine given that:
- The exact mass of 35Cl is 34.9689 and the relative abundance is 75.53%
- The exact mass of 37Cl is 36.9659 and the relative abundance is 24.47%
Is this the answer that you expected?
- What is the Atomic Weight of ruthenium given the following information from WebElements:
| Isotope | Abundance | Exact Mass |
|---|
| 96Ru | 5.54% | 95.907599 |
| 98Ru | 1.87% | 97.905287 |
| 99Ru | 12.76% | 98.9059389 |
| 100Ru | 12.60% | 99.9042192 |
| 101Ru | 17.06% | 100.9055819 |
| 102Ru | 31.55% | 101.9043485 |
| 104Ru | 18.62% | 103.905424 |
Is this the answer that you expected?
- Using a
blank periodic table, fill in the atomic number, name, atomic symbol, and atomic weight of the first 36 elements. Also label the noble gases, the halogens, the alkali metals, the metals, and the non-metals.
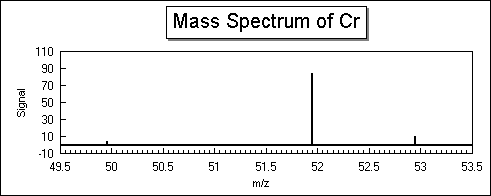
- Use the mass spectrum for chromium given below to determine the atomic weight.
Please send comments or suggestions to svanbram@science.widener.edu
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
© copyright 1996, S.E. Van Bramer
This page has been accessed
times since 1/5 /96 .
Last Updated: Saturday, May 18, 1996
