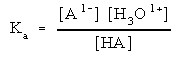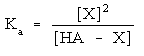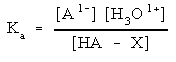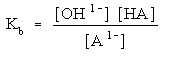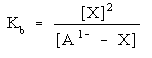Since it is a strong acid it dissociates completely, you will not have any HCl. Cl1- is not going to do anything in an acid base reaction. It is a spectator ion so leave it out. What you need is to figure out the [H3O1+] (ie: the concentration of hydronium ions). Remember concentration is moles over liters.
Since it is a strong base it dissociates completely. You will not have any NaOH. Na1+ is not going to do anything in an acid base reaction. It is a spectator ion so leave it out. What you need to do is figure out the [OH1-] (ie: the concentration of hydroxide ions). Remember concentration is moles over liters.
Then write out the equilibrium expression:
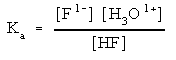
You will need to use this expression to determine what concentration each species is at when the system is at equilibrium. Exactly how to solve this depends upon what other species are present. More on that coming up.
You need to do this so that you can decide what will happen in solution. Almost everyone in the class missed one or more problem because they failed to recognize a salt, and then see what it will do in solution. After you write the salt out as ions, look at the ions to see if you recognize any of them as a weak acid or a weak base. Or the conjugate acid or base. Anything with a Ka or Kb. After you recognize it as an acid or base, write the appropriate reaction.
This step can be a bit tricky. For the example here F1- is the conjugate base of a weak acid. That means that it would like to gain a proton (H1+) from something. The reaction that you write depends upon what species are present. The idea here is to think about what the strongest acid is. If only the salt is present, the strongest acid is water.
But, If any acid is present you can write a different reaction.
Notice that this is like the acid dissociation of HF shown above. Since HF is a weak acid, F1- is also a weak base. That means you need to use an equilibrium expression to solve for the concentration of each species. We will deal with the equilibrium expressions below.