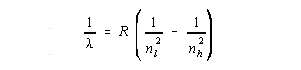
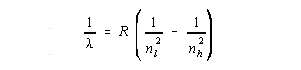
| Quantum Number | Name | Value | Meaning |
| n | Principal Quantum Number | Integer (1, 2, 3, .). | Shell |
| l | Angular Momentum | 0 to n-1 | Type of orbital (s, p, d, f) |
| ml | Magnetic Quantum Number | -l to +l | Orientation |
| ms | Spin | + or - 1/2 | Electron Spin |
Please send any comments, corrections, or suggestions to svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 1:59:43 PM