Chapter 10 Lecture Problems
10.1 Artificial Sweeteners: Fooled by Molecular Shape
10.2 VSEPR Theory: The Five Basic Shapes
10.3 VSEPR Theory: The Effect of Lone Pairs
10.4 VSEPR Theory: Predicting Molecular Geometries
10.5 Molecular Shape and Polarity
10.6 Valence Bond Theory: Orbital Overlap as a Chemical
10.7 Valence Bond Theory: Hybridization of Atomic Orbitals
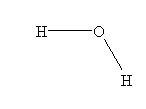
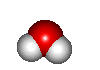
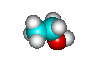
- H2O
- C2H6
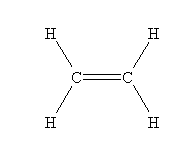
- C2H4
- PDB File
 13K
13K
- Sigma Bonds (
Internet © Saunders, 1997)
- Pi Bonds (
Internet © Saunders, 1997)
- All Bonds (
Internet © Saunders, 1997)
- Butane, rotation about single bond (
Internet © Saunders, 1997)
- 2-butene, rotation about double bond (
Internet © Saunders, 1997)
- C2H2
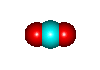
- CO2
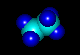
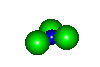
- NH3
- NH4+
- BH3
- BCl3
- N2
- CH3CH2OH
- CH3NH2
10.8 Molecular Orbital Theory: Electron Delocalization
This page is maintained by
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
Please send any comments, corrections, or suggestions to
svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 2:10:36 PM

 13K
13K
 13K
13K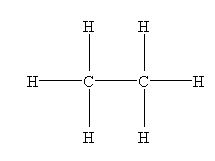
 13K
13K
 13K
13K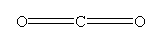
 13K
13K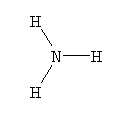
 13K
13K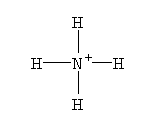
 13K
13K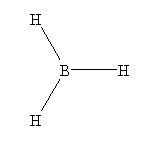
 13K
13K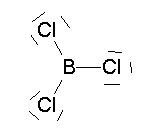
 13K
13K
 13K
13K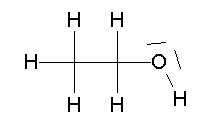
 13K
13K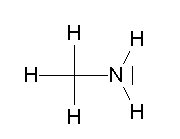
 13K
13K