- Data from Zumdahl
time NO2 NO O2 0 0.0100 0 0 50 0.0079 0.0021 0.0011 100 0.0065 0.0035 0.0018 150 0.0055 0.0045 0.0023 200 0.0048 0.0052 0.0026 250 0.0043 0.0057 0.0029 300 0.0038 0.0062 0.0031 350 0.0034 0.0066 0.0033 400 0.0031 0.0069 0.0035 - Graph concentration vs time for NO2
- Calculate concentration of other species and graph
- Calculate average rate
- from 0 to 50
- from 200 to 250
- Completed Spreadsheet
- Compare rates for each species
- CD-ROM 15-2 side bar (Bleach and dye)
- Surface Area. ( internet © Saunders, 1997)
- Concentration. CD-ROM, 15-4 (
internet © Saunders, 1997)
- Mg (s) + 2 HCl (aq) -> H2 (g) + Mg2+ + 2 Cl1- (aq)
- 2 N2O5 -> 4 NO2 + O2 (Initial Rate vs concentration)
The Rate Law: The Effect of Concentration on Reaction Rate
- The rate law
- Expression:
for a reaction: aA + bB -> cC + dD
rate is rate = k [A]x [B]y
- Expression:
- Discuss units for kinetics problems (Handout).
- Method of Initial Rates:
Determine rate law from initial rates or rates at different concentrations. (Lecture Problems).
- Derive Relationship
ln(Rate1/Rate2) = x ln(A1/A2) Where:- Rate1 is the rate under the first conditions.
- Rate2 is the rate under the second conditions.
- A1 is the concentration of A for the first conditions.
- A2 is the concentration of A for the second conditions.
- x is the reaction order for A.
- For the reaction:
NH4+(aq) + NO2-(aq) -> N2(g) + 2 H2O (l) With the following experimental data.
Experimental Data [NH41+] (M) [NO21-] (M) Rate (M s-1 0.100 0.005 1.35*10-7 0.100 0.010 2.75*10-7 0.200 0.010 5.4*10-7 - Determine the order of a reaction
- Determine k for reaction:
- Write the rate law
- Use the rate law to calculate the rate for new concentrations.
The Integrated Rate Law: The Dependence of Concentration on Time
(Lecture Problems)- Plot of concentration vs time, and rate vs time
- Units for axis
- Units for area under curve
- Integration of rate equation
- Derive 1st order integrated rate equation
- Use the integrated rate equation to find the concentration at time t for:
C2H5Cl -> C2H4 + HCl at 700 K, k = 2.50*10-3 min-1 - If the initial concentration of C2H5Cl is 3.45 atm, how long does it take for the partial pressure of C2H5Cl to drop to 1.00 atm?
- What is the partial pressure of C2H5Cl after six hours?
- How long does it take for the C2H5Cl concentration to drop to 1/2 the initial concentration?
- How long does it take for 99% of the C2H5Cl to react?
- Graph Concentration vs time from integrated rate equation (overhead).
Rate laws, integrated rate equation, and units
| Reaction Order | Rate | Rate Law | Integrated Rate Equation | Linear Plot | Slope | Units for k |
| 0 | [delta] concentration ----------------------- [delta] time |
rate = k | [A]0 - [A]t = kt | [A] vs time | -k | concentration time-1 mol liter-1 sec-1 pressure time-1 |
| 1 | [delta] concentration ----------------------- [delta] time |
rate = k [A] | ln ([A]t/[A]0) = - kt | ln [A] vs time | -k | sec-1 |
| 2 | [delta] concentration ----------------------- [delta] time |
rate = k [A]2 rate = k [A] [B] |
(1/[A]t) - (1/[A]0) = kt | (1/[A]) vs time | k | concentration-1 time-1 liter mole-1 sec-1 pressure-1 time-1 |
Graphing Kinetics Data
Graphing Kinetics Data (pdf)
The Effect of Temperature on Reaction Rate
- Collision Theory
- Collision Energy NO + O3 -> NO2 + O2
(
internet© Saunders, 1997)
- Orentation NO + O3 -> NO2 + O2
( internet© Saunders, 1997)
- Successful Reaction NO + O3 -> NO2 + O2
( internet© Saunders, 1997)
- Energy Diagram (Overhead)
- Reaction Energy Diagram F1- + CH3Cl -> CH3F + Cl1- (
internet© Saunders, 1997)
- Temperature and reaction rate, bleach demo (
internet © Saunders, 1997)
- Concentration and reaction (collision) rate).
- Collision Energy NO + O3 -> NO2 + O2
(
internet© Saunders, 1997)
- The Arrhenius Equation
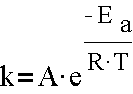
- Graphing ln(k) vs 1/T, idea of linear relationships
- Derive relationship
- Show graph on Lotus
- Calculations with the Arrhenius equiaton
- Given A and Ea , determine k at T
- Find change in rate at two T's from Ea
- Determine Ea from k at two T's
- Arrhenius Plot to find Ea
- Given A and Ea , determine k at T
- (Lecture Problems)
Reaction Mechanisms
Catalysis
- H2O2 Decomposition ( internet© Saunders, 1997)
- Catalysis reaction steps
- Given the reaction steps:
- NO + O3 -> NO3 + O (slow)
NO3 + O -> NO2 + O2 (fast)NO + O3 -> NO2 + O2 (overall)
- And an additional step:
- Combines as:
- Which species is a catalyst?
- Which species is an intermediate?
- How does this catalyst effect the rate of the reaction
NO2 + O -> NO + O2
NO + O3 -> NO2 + O2
NO2 + O -> NO + O2O3 + O -> 2 O2
- Catalysis Energetics and Reaction Rates. (Lecture Problems)
For the ovrall reaction: O3 + O -> 2 O2
- For the catalyzed reaction Ea = 11.9 kJ
- For the uncatalyzed reaction Ea = 14.0 kJ
- Draw an energy level diagram
- What is the rate constant at 298 K for each mechanism?
- For the catalytic destruction of O3 by Cl Ea = 2.1 kJ, what would this rate constant be at 298 K?
This page is maintained by
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
Please send any comments, corrections, or suggestions to svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 1:59:43 PM