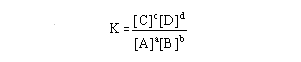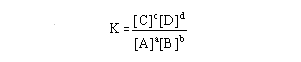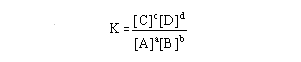Chapter 14 Outline
14.1 Fetal Hemoglobin and Equilibrium
14.2 The Concept of Dynamic Equilibrium
14.3 The Equilibrium Constant (K)
The general equation for equilibrium constant (K)
For a reaction aA + bB <--> cC + dD
14.4 Expressing the Equilibrium Constant in Terms of Pressure
When equlibrium is calculated using concentration, use Molarity. When equlibrium is calculated using pressure you must be consistent about which units are used. The textbook says pressure should be in atm, but it can be in any unit as long as you are consistent. You can convert between Kc and Kp using the ideal gas law and the change in moles of gas from reactant to product so that:
Kp = Kc(RT)delta_n
14.5 Heterogeneous Equilibrium: Reactions Involving Solids and Liquids
Solids and pure liquids have constant concentration - as long as they are present, so they are not included in the equlibrium expression - they are already part of the constant.
14.6 Calculating the Equilibrium Constant from Measured Equilibrium Concentrations
The general equation for equilibrium constant (K)
For a reaction aA + bB <--> cC + dD
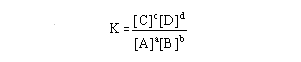
14.7 The Reaction Quotient: Predicting the Direction of Change
q is calculated using the same equation as K - but it is NOT at equlibrium. You can use this to predict the direction of the reaction, since q will go to K as the system goes to equlibrium.
14.8 Finding Equilibrium Concentrations
Learn how to use the Initial, Change, Final table. You MUST be able to solve these problems.
14.9 Le Chatelier's Principle: How a System at Equilibrium Responds to Disturbances
This principle lets you predict the effect of changing concentration, temperature, and volume on an equlibrium system.
This page is maintained by
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
Please send any comments, corrections, or suggestions to
svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 2:11:17 PM