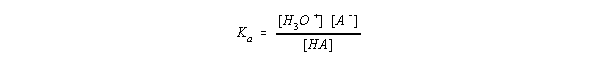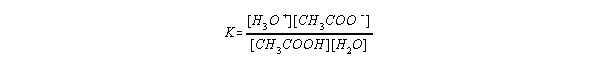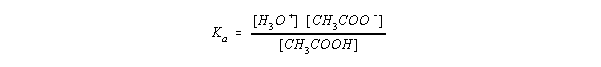Chapter 15 Lecture Outline
15.3 Definitions of Acids and Bases
The Arrhenius Definition
Bronsted-Lowry Acid-Base Reactions
- General Equation
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| HA | + | B | --> | A- | + | HB+ |
- Strong Acid and Strong Base
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| HCl(aq) | + | NaOH(aq) | --> | Cl- | + | Na1+ | + | H2O (aq) |
- Weak Acid and Weak Base
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| CH3COOH(aq) |
+ |
H2O(l) |
<--> |
CH3COO-(aq) |
+ |
H3O+(aq) |
- Autoionization of water:
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| H2O(l) |
+ |
H2O(l) |
<--> |
OH-(aq) |
+ |
H3O+(aq) |
- Polyprotic Acid
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| H2SO4 |
+ |
H2O(l) |
--> |
HSO41- |
+ |
H3O+(aq) |
| HSO41- |
+ |
H2O(l) |
<--> |
SO42-
|
+ |
H3O+(aq) |
15.4 Acid Strength and the Acid Ionization Constant (Ka)
- Binary acids X-H bond strength:
- Oxyacids, more O, stronger acid (Electron withdrawing by X in X-OH)
| Acid | Formula | Acid Dissociation Constant |
| hypochlorous acid | HOCl | Ka = 10-8 |
| chlorous acid | HOClO | Ka = 10-2 |
| chloric acid | HOClO2 | Ka = 101 |
| perchloric acid | HOClO3 | Ka = 108 |
- Metal hydroxides, X is electroposative
- NaOH strong base
- Be(OH)2 amphoteric
- Carboxylic acids R-COOH
15.5 Autoionization of Water and pH
| Acid | | Base | | Conjugate
Base of HA | | Congugate
acid of B |
| H2O(l) |
+ |
H2O(l) |
--> |
OH-(aq) |
+ |
H3O+(aq) |
- The equilibrium concentrations can be described with a constant K for the autoionization of water:
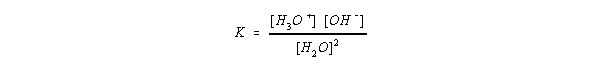
- This is usually rearranged since [H2O] is essentially constant. and
given as Kw:
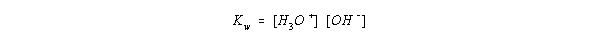
- At 25 C, Kw = 1.0 * 10-14
- Calculate [OH-] and [H3O+]
- 0.1 M HCl
- 0.1 M NaOH
- pH and pOH
- pH = - log[H3O+]
- pOH = - log[OH-]
- pH + pOH = 14 (Show algebra)
15.6 Finding the [H3O+] and pH of Strong and Weak Acid Solutions
- Generic acid reaction
- HA + H2O <--> A- + H3O+
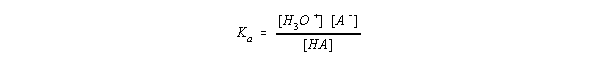
- Look up Ka in table
- Acetic acid:
- For the reaction CH3COOH + H2O <--> CH3COO- + H3O1+
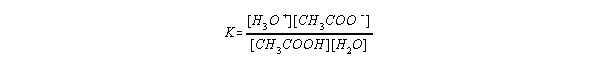
- Since [H2O] is constant, this is typically expressed as Ka
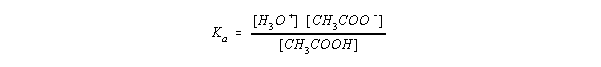
- Ka = 1.8 x 10-5
- Calculating pH (Given Solution concentration)
| Solution | [H3O+] | pH | [OH-] | pOH |
| 10 M HCl | 101 | -1 | 10-15 | 15 |
| 0.1 M HCl | 10-1 | 1 | 10-13 | 13 |
| 10-8 M HCl | 10-8 | 8 | 10-6 | 6 |
| 10 M NaOH | 10-15 | 15 | 101 | -1 |
| 0.1 M NaOH | 10-13 | 13 | 10-1 | 1 |
| 10-8 M NaOH | 10-6 | 6 | 10-8 | 8 |
- Note: pH + pOH = 14
- Note: 10-8 M HCl and NaOH make no sense, why?
- Significant figures and logs
- Calculate [H3O+] and [OH-] given pH or pOH
- Vinegar pH=2.9
- ammonia pH = 11.5
Example Problems: Solving a Weak Acid Equlibrium System
- For a nitrous acid (Ka = 4.5 x 10-4) solution find the pH and pOH for initial nitrous acid concentrations of
- 1.0 M
- 0.001 M
- 10-8 M.
- Solutions
- Acetic Acid Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of acetic acid is added to 250.0 mL of water.
- Solution
- Nitrous Acid Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of nitrous acid is added to 250.0 mL of water.
- Solution
- Nitric Acid Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of nitric acid is added to 250.0 mL of water.
- Solution
15.7 Base Solutions
- Ka =
- Kb =
- Kw =
- Kw = Ka * Kb
15.8 The Acid-Base Properties of Ions and Salts
- Chemical System
- Reaction #1 CH3COOH + H2O <-> CH3COO- + H3O+
- Reaction #2
- step 1: NaCH3COO --> Na1+ + CH3COO1-
- step 2:
CH3COO1- + H2O <-> CH3COOH + OH-
- TOTAL rxn: 2 H2O <-> H3O+ + OH-
- Equlibrium for reaction #1 is Ka for CH3COOH
- For a 0.100 M CH3COOH at equilibrium, calculate Ka if at equlibrium [H3O] = 1.318*10-3 M:
| |
CH3COOH |
H2O |
<--> |
CH3COO- |
H3O+ |
| Initial (M) |
0.100 |
|
|
0 |
0 |
| Change (M) |
-1.318*10-3 |
|
|
+1.318*10-3 |
+1.318*10-3 |
| Final (M) |
0.0987 |
|
|
1.318*10-3 |
1.318*10-3 |
- Flow Chart
- Examples. What happens when...
- sodium acetate dissolves in water.
- potassium nitrate dissolves in water.
- ammonium chloride dissolves in water.
- sodium fluoride dissovles in water.
- potassium hydroxide dissolves in water.
Example Problems
- Sodium Acetate Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of sodium acetate is added to 250.0 mL of water.
- Solution
- Ammonium Chloride Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of ammonium chloride is added to 250.0 mL of water.
- Solution
- Sodium Nitrite Equlibrium
- Calculate the H3O+, OH-, acid concentration, base concentration, pH and pOH when 1.00 g of sodium nitrite is added to 250.0 mL of water.
- Solution
15.9 Acid Strength and Molecular Structure
15.10 Lewis Acids and Bases
This page is maintained by
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
Please send any comments, corrections, or suggestions to
svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 1:59:43 PM
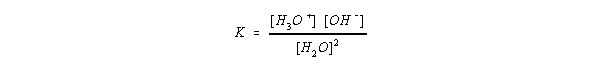
![]()
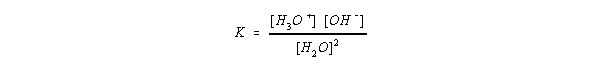
![]()