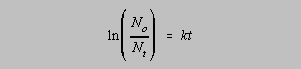Chapter 19 Lecture Outline
Diagnosing Appendicitis
Types of Radioactivity
- Nuclear Particles
| Name | Symbol | Symbol | Mass | Charge |
| Alpha | [alpha] | 4He | 4 | +2 |
| Beta | [beta] | 1-e | 0 | -1 |
| Gamma | [gamma] | | 0 | 0, radiation (light) |
| Neutron | n | | 1 | 0 |
| Proton | p | 1H | 1 | +1 |
| Positron | +1e | +[beta] | 0 | +1 |
- Radioactivity and
reactions
- Alpha emission: 238U -> 234Th + 4He
- Beta Emission:
- 14C -> 14N + -1e
- 1n -> 1p + -1e (neutron -> proton + electron)
- Positron Emission
- 11C -> 11B + +1e
- 1p -> 1n + +1e (proton -> neutron + positron)
- Electron Capture
- 40K + -1e -> 40Ar
- 1p + -1e -> 1n (proton + electron -> neutron)
The Valley of Stability: Predicting the Type of Radioactivity
Nuclear Stability. Likelihood that isotope is radioactive depends upon:
- >83 protons are unstable
- Magic Numbers 2, 8, 20, 28, 50, 82 are stable. If magic number of both protons and
neutrons, then "double magic"
- Even numbers of neutrons or protons are stable.
- Both even, most stable
- One even one odd, less stable
- both odd, least stable
- Belt of stability
- If above, then decay to convert a neutron to a proton.
- If below, then positron emission (or electron capture) to convert a proton to a
neutron.
The Kinetics of Radioactive Decay and Radiometric Dating
- Half life of radioactive isotopes
- Radioactive decay is first order
- First order rate law
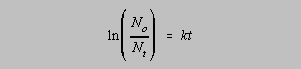
- For half life t1/2 = 0.693/k
- Amount remaining after time t
- Uses of Radioactivity (See Mathcad)
- 238U Dating (Age of Rock Problem from Page 907)
- 238U -> 206Pb t1/2 = 4.5*109 years
- 40K Dating
- 40K + -1e -> 40Ar t1/2 = 1.3x109 years
- 14C Dating
- 14N + 1n -> 14C + 1H
- 14C -> -1e + 14N t1/2 = 5730 years
- Shroud of Turin (Dated at 1300 AD). Determine 14C present. What would it be if 0 AD?
The Discovery of Fission: The Atomic Bomb and Nuclear Power
Converting Mass to Energy: Mass Defect and Nuclear Binding Energy
Nuclear Fusion: The Power of the Sun
The Effects of Radiation on Life
Radioactivity In Medicine
Production of New Nuclei
- Particle accelerators
- Cyclotron (Figure 24.7, page 903)
- Linear Accelerator (Figure24.8, page 903)
- Induced Radioactivity (Making a radioactive isotope)
- 230Th + 1H --> 223Fr + 2 4He
- Neutron Bombardment (Use fast neutrons to prepare isotopes)
- 58Fe + 1n -> 59Fe
- 59Fe -> 59Co + -1e
- 59Co + 1n -> 60Co (Used for radiation treatment of cancer)
This page is maintained by
Scott Van Bramer
Department of Chemistry
Widener University
Chester, PA 19013
Please send any comments, corrections, or suggestions to
svanbram@science.widener.edu.
This page has been accessed
times since 5/30/97.
Last Updated Friday, May 25, 2001 1:59:43 PM